New findings from the Antimicrobial Discovery Center, Northeastern University based on funding from Global Lyme Alliance
Xiaoqian Wu, Bijaya Sharma, Samantha Niles, Kathleen O'Connor, Rebecca Schilling, Nicole Matluck, Anthony D'Onofrio, Linden T. Hu and Kim Lewis DOI: 10.1128/AAC.01201-18 August 2018
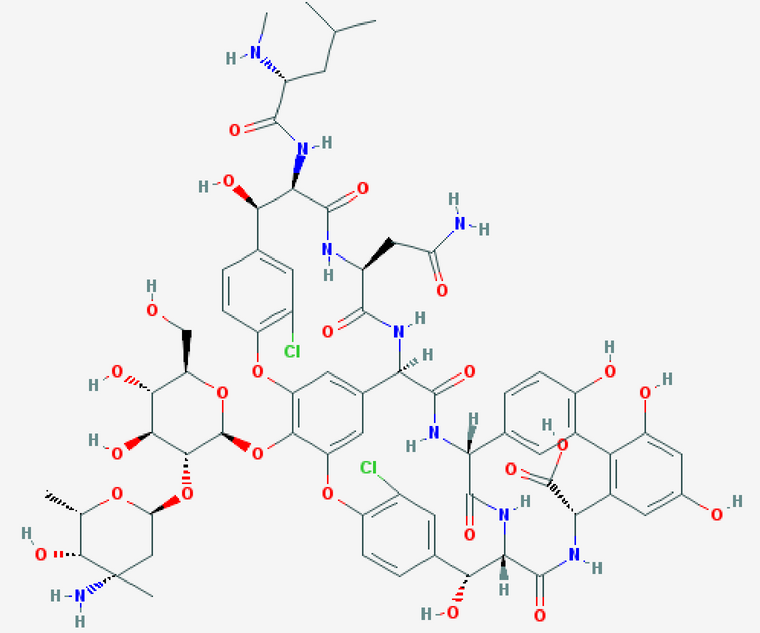
ABSTRACT Borrelia burgdorferi is the causative agent of Lyme borreliosis. Antibiotic therapy of early acute infection is effective for most patients, but 10-20% go on to develop Post-Treatment Lyme Disease Syndrome. The nature of PTLDS remains unknown, but currently approved antibiotics for treatment of Lyme disease do not appear to impact these symptoms after they have developed. We reason that minimizing the time the pathogen interacts with the host will diminish the probability of developing PTLDS, irrespective of its nature. This calls for an efficient eradication of the pathogen during acute infection. In search of a superior killing antibiotic, we examined approved antibiotics for their ability to kill B. burgdorferi. Vancomycin (pictured) proved more effective in killing the pathogen in vitro than ceftriaxone, the standard of care for disseminated B. burgdorferi infection. Both compounds were also the most effective in killing stationary phase cells. This is surprising, given that inhibitors of cell wall biosynthesis are known to only kill growing bacteria. We found that peptidoglycan synthesis continues in stationary cells of B. burgdorferi, explaining this paradox. A combination of vancomycin and gemifloxacin sterilized a stationary phase culture of B. burgdorferi. Examination of the action of antibiotics in immune-deficient SCID mice showed that doxycycline, a standard of care for uncomplicated acute infection, did not clear the pathogen. By contrast, both ceftriaxone and vancomycin cleared the infection. A trial examining early use of more potent antibiotics on development of PTLDS may be warranted. Access full report
Pictured: A structure of vancomycin

